Get in touch with our highly skilled team to discuss your surgical instrument requirements
- British Manufacturer of Surgical Instruments
- |
- Established Supplier with Over 35 years Experience
- |
- British Instrument, Scope & Power Tool Repair

Bone Pins & Skeletal Wires
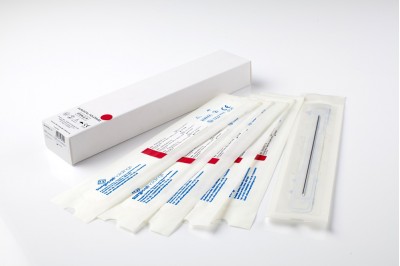
At Surgical Holdings, we provide a range of bone pins and skeletal wires for internal fixation during orthopaedic surgery. We manufacture a range of Wires and Pins including Sterile Kirschner Wires, Arthrodesis Wires, Steinmann and Pins and Denham Threaded Steinmann Pins for internal fixation during orthopaedic surgery. We are accredited to sell these products (Class IIb product) into Europe, and the UK through the NHS and Private Hospitals by SGS (View certificate).
- These products are only to be implanted by a qualified medical practitioner using compatible CE marked instruments/Devices.
- Skeletal wires should only be used for fusion, skeletal traction, and fixation (to fixate bones while they are healing) in the areas of the body for which they were intended, namely hands and feet.
- Bone pins and skeletal wires are non-permanent implants, they should be removed after 120 days, and SHOULD NEVER BE REUSED. The skeletal wires should never be misused in any way.
All Implants are manufactured as per the relevant international standard for material, ISO 5838-1: 2013. A full listing of standards we manufacture to is shown below:
ISO 5832-1:2016 Metallic materials for surgical implants. Specification for wrought stainless steel.
|
BS 3531 Part 6.2 ISO 5838-2 |
1991 1991 |
Implants for osteosyntheses. Specification for dimensions of Steinmann skeletal pins |
|
BS 3531 Part 6.3 ISO 5838-3 |
1994 1993 |
Implants for osteosyntheses. Skeletal pins and wires. Specification for Kirschner skeletal wires |
|
|